Addressing Aspiration Risk at Its Source
Pulmonary aspiration of gastric contents remains a serious and unresolved risk in anesthesia and critical care—particularly in urgent, emergent, or physiologically unstable patients where traditional safeguards are difficult to apply. Aspiration can lead to pneumonia, hypoxemia, acute respiratory distress syndrome, and even death. While the risk is widely recognized, current strategies rely primarily on indirect measures such as fasting protocols, patient positioning, airway timing, and pharmacologic acid suppression. These approaches simply mitigate the consequences without reliably preventing aspiration.
As a result, concern for aspiration frequently drives delays in care, procedural deferral, or prolonged observation, especially in high-risk patients. Despite decades of clinical awareness, there remains no widely adopted method that directly addresses the mechanical movement of gastric contents toward the esophagus at the moment that risk is highest. Further, with increasing use of medications(GLP-1’s) and conditions(obesity, diabetes, etc) that slow gastric emptying, clinicians are only facing this threat more often. This gap leaves clinicians balancing safety concerns against time-sensitive decision-making, often without a clear, evidence-based tool designed for this purpose.
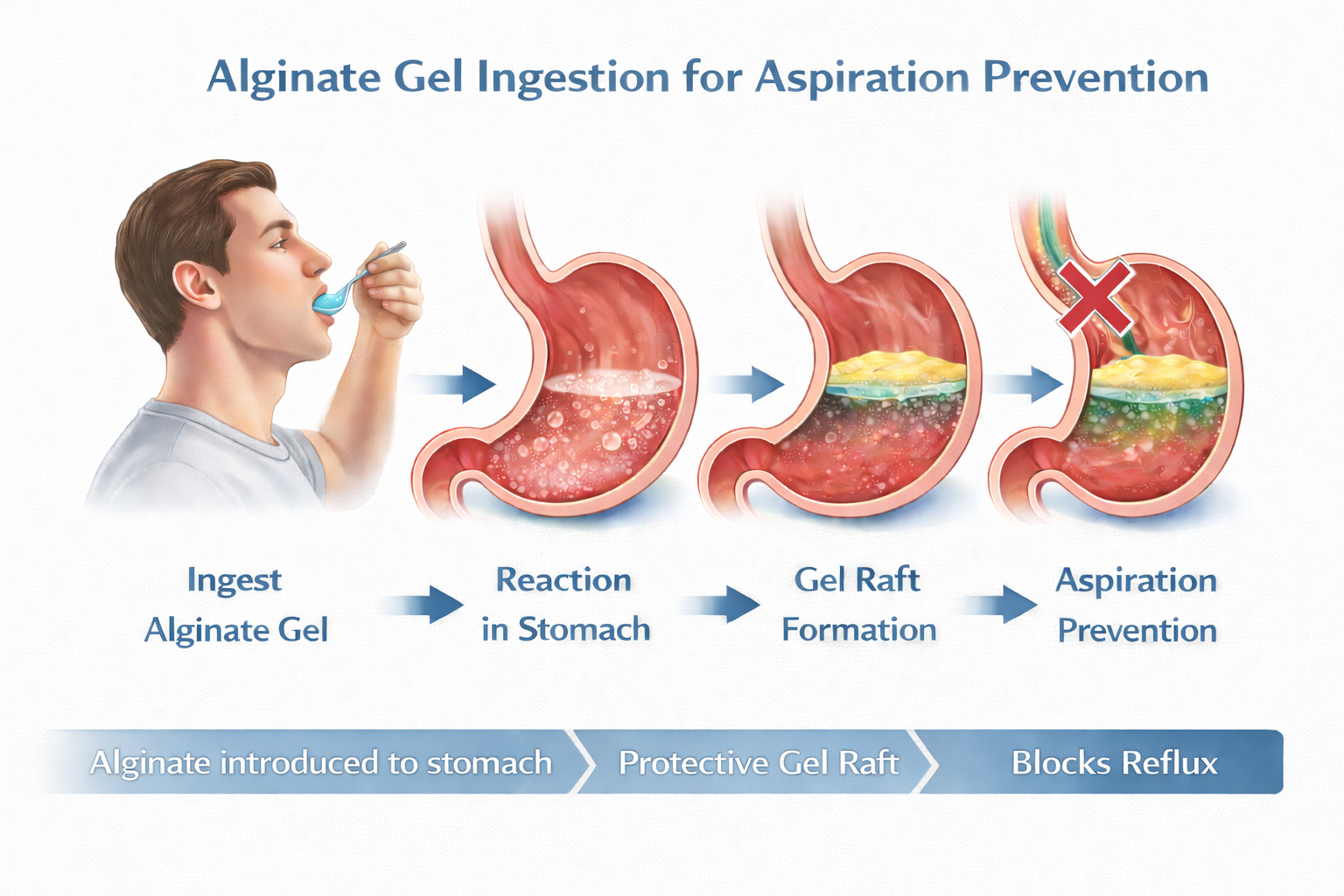
Our mission is to address this unmet need by exploring a localized, barrier-based approach focused on the stomach–esophagus interface itself—one that acts where aspiration risk originates rather than downstream. This focus naturally leads to materials capable of forming reliable, localized barriers under gastric conditions. This is where alginates come in.
Alginates
From Proven Material to Purpose-Built Platform
Alginates are naturally derived polysaccharides isolated from brown seaweed and have been used for more than a century across pharmaceutical, medical, and industrial applications. Their enduring value stems from a unique combination of biocompatibility, predictable gel-forming behavior, and an extensive history of safe human exposure. Commercially available alginates have been incorporated into a wide range of products intended for human use, resulting in a well-established safety profile supported by decades of real-world experience and scientific study.
We chose alginates as the foundation of our platform because their intrinsic behavior aligns naturally with our vision for a simple, elegant solution to a complex clinical problem. In gastric conditions, alginates inherently form cohesive, buoyant gels through localized ionic interactions. This behavior enables a physical, barrier-based approach that operates precisely where it is needed while remaining localized and non-systemic. By starting with a material that is deeply understood, widely trusted, and commercially established, we are able to focus on purposeful formulation innovation—enhancing strength, durability, resistance to fracture and functional performance—to unlock new possibilities beyond traditional applications. This intentional evolution of a proven material forms the basis of our platform and underpins our approach to addressing clinically meaningful risk through intelligent material design.
Our Innovation
Our technology platform builds on the known gel-forming properties of alginates while introducing formulation innovations designed to enhance gel strength, persistence, and structural integrity under gastric conditions. Throughout in-vitro testing, our alginate formulation has demonstrated the ability to form a more cohesive and durable gel structure compared with conventional alginate systems. These properties are intended to support sustained localization at the gastric fluid interface over clinically relevant timeframes and physiologic conditions. Under controlled laboratory conditions, this enhanced gel structure has demonstrated a marked reduction in the upward movement of gastric contents into a esophagus model, including under simulated supra-physiologic intra-abdominal pressures, supporting the feasibility of a barrier-based approach to reflux mitigation and thus reduction in aspiration incidence. By optimizing a well-characterized material rather than introducing a novel systemic agent, our platform emphasizes predictable behavior, scalability, and a focused development pathway centered on physical mechanism rather than pharmacologic activity.